A
lthough research regarding skin aging mainly focuses on aesthetic aspects, skin alteration caused by aging is more than a merely cosmetic issue [1-5]. Cutaneous aging is a complex biological phe-nomenon consisting of two distinct components: intrin-sic genetically determined aging processes and extrinsic aging due to exposure to environmental factors, such as UV radiation and pollution [6-8].
Physiological changes in aged skin include structural and biochemical alterations, as well as changes in neurosen-sory perception, permeability, response to injury, and repair capability, which increase in some skin diseases, among them, skin cancer. In 2015, the World Health Organization World Report on Aging and Health emphasized the necessity to develop strategies dealing with aging, and the skin was identified as a domain that deserves greater attention [9].
In 2013, Lopez-Otin et al. first proposed the concept of biological geriatric assessment (BGA). This was updated in 2023 and includes 12 hallmarks considered as the denominators of aging including that of skin. These include genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis (protein homeostasis), disabled macro-autophagy, dysregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communica-tion, chronic inflammation, and dysbiosis.
The proteome includes all proteins present in an organism or cell, and control of the proteome is referred to as “proteome homeostasis” or “proteostasis”. The proteome plays an essential role in all life forms since proteins, directly or indirectly, carry out all life func-tions. In the human body, proteins represent the second most abundant constituent after water [10]. However, to date, only modest attention has been devoted to proteome alterations in studies on aging.
In a recent publication, a focus was given to the role of the proteome in aging, including that in the skin as well as age-related diseases [11]. The present review provides a focus on skin aging with relevant information about the proteome, and moreover details arguments in favour of the proteome as a potential central target in the strategy of prevention and management of skin aging that encompasses other well-known pathways of skin aning.
Methodology
A comprehensive review of 87 articles available from PubMed was conducted. Focus was given on aging, the proteome, proteostasis, the proteasome, protein carbon-ylation and BGA, as well as its hallmarks: genomic insta-bility, telomere attrition, epigenetic alterations, loss of proteostasis, dysregulated nutrient sensing, mitochon-drial dysfunction, cellular or immune senescence, stem cell exhaustion, and altered intercellular communication in dermatology using the above key words, either alone or combined with each other.
Results
In the skin, as in any other body tissue or cell, proteins are involved in each of the aging hallmarks stated by Lopez-Otin et al., consistent with the dependence of nearly all life functions on dedicated protein activities [12, 13].
Protein turnover
Protein turnover involves protein degradation (proteol-ysis) and resynthesis to maintain and regulate protein activity and remove abnormal (most frequently caused by oxidative damage), dysfunctional or cytotoxic proteins. In young tissues, damaged proteins are effi-ciently eliminated by selective proteolysis, thus avoiding dysfunction and cytotoxic effects of oxidized small aggregates, called “oligomers” [14, 15].
Natural protein degradation is performed via the ubiq-uitin-proteasome system (UPS), an enzymatic complex dedicated to protein degradation which is present in all cells [16-19].
Protein quality control (PQC), involving multiple chap-erones and degradative pathways, identifies and elimi-nates abnormal and misfolded proteins susceptible to carbonylation, that are deleterious to cells and tissues [20]. If PQC fails, then protein aggregation may occur. A certain number, but not all, of these aggregations are eliminated via autophagy [5, 21-23]. Thus, during aging, these protein aggregates accumulate in tissues, promoting the development of chronic inflammation.
In the skin, protein damage mostly results from external stimuli, including exposure to oxidants present in air pollution, ozone, chemical agents and radiation, such as UV light [24-29]. Moreover, protein damage can be caused through internally generated oxidants and reac-tive oxygen and nitrogen species, which are produced during metabolism or immune responses [30, 31]. Finally, protein damage can occur due to transcriptional or translational errors, since misfolded proteins become sensitive to carbonylation [32, 33]. Such biosynthetic errors can occur naturally, but become more prevalent with age due to a deficient feedback loop between accu-mulating damage and transcriptional and translational efficacy and fidelity [34].
Skin aging is a permanent process
Skin and tissue aging via protein damage is mainly caused by protein glycation (figure 1A), carbon-ylation (figure 1B) and, to a lesser extent, through The accumulation of advanced glycation end products (AGEs), protein carbamylation and protein carbonyla-tion (PC) products has been causally associated with skin aging [35-37]
Protein glycation
During protein glycation, AGEs are derived from the modification of proteins or lipids that are glycated by reducing sugars [38-40]. They modify cell structure and extracellular matrices, which also results in the release of free radicals [41]. AGE accumulation in the dermis results in yellow, discoloured skin. Collagen cross‐links reduce flexibility and mobility, resulting in the loss of skin elasticity [41-46]. AGEs also induce strong nuclear factor‐κB activation and cause inflammation in the skin via pro‐inflammatory cytokines [44, 47].
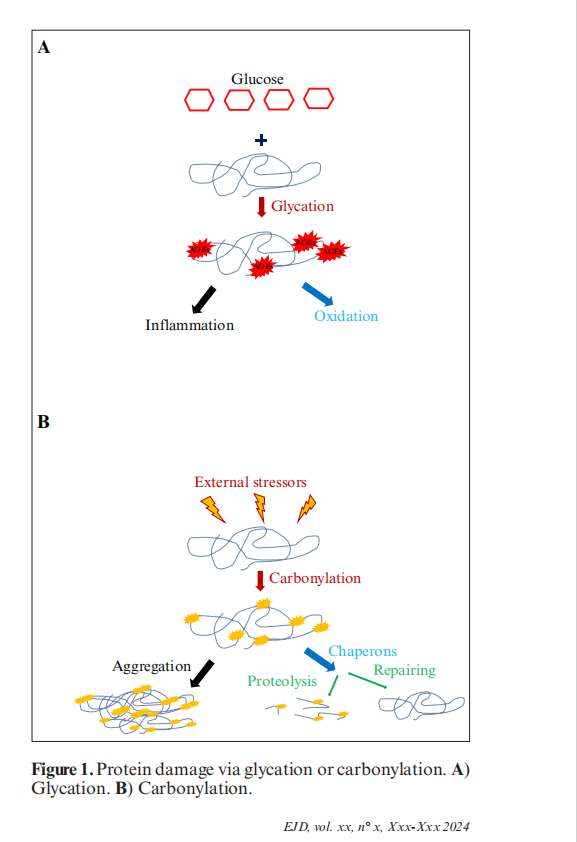
Unlike irreparable carbonylation, glycation is subject to efficient repair by Parkinsonism-associated Protein DJ-1/Park7 (DJ-1/PARK-7 deglycase) [48]
Protein carbonylation (PC)
PC forms reactive ketones or aldehydes [49]. It causes irreversible oxidative damage, frequently resulting in the loss of protein function and protein aggregation [50]. In the skin, PC results in several distinguishable clinical changes. Transepidermal water loss is increased in the stratum corneum [51-53]. In the supra-basal epidermis, the accumulation of carbonylated keratins disrupts light transmission, which alters subjectively perceived skin radiance and homogeneity of skin complexion [54, 55]. Carbonylated proteins alter the dermis by degrading collagen and elastin. PC is associated with an alteration of fibroblasts, as well as changes in the expression of metalloproteases, such as MMP-1, and the development of chronic inflammation involving IL-8 [56-59]
Protein carbamylation
Protein carbamylation is a non-enzymatic modification which involves the binding of isocyanic acid to protein functional groups. This reaction alters the structure and function of proteins, resulting in protein damage and chronic inflammatory diseases [37].
Proteins as markers of skin aging
A recent quantitative proteomic analysis identified a total of 95 differentially expressed proteins (DEPs) in aged skin, compared to young skin. An enrichment anal-ysis of 57 upregulated and 38 downregulated proteins showed different functional clusters related to immunity and inflammation, oxidative stress, biosynthesis and metabolism, proteases, cell proliferation and differentiation, as well as apoptosis [60]. The most rele-vant up-regulated proteins in aged skin samples were galectin-3-binding protein and demicidin (both involved in the immune and inflammatory response), serpin B6 (a protease inhibitor), mucin-like protein 1 (a signal transduction related protein), melanotransferrin (a metal ion-containing protein), and the following proteins involved in cell apoptosis, as well as cell proliferation and differentiation: glypican-1, prelamin-A/C, syntening-2 and annexin A2. The most relevant down-regulated proteins were keratin type I cytoskeletal 15 and 39, keratin type II cytoskeletal 72, as well as keratin type I cuticular Ha5 and keratin type II cuticular Hb2. Other down-regulated proteins included cyto-plasmic histidine-tRNA ligase and cytoskeleton-related protein and extracellular matrix protein 1. Moreover, glyceraldehyde-3-phosphate dehydrogenase, that regu-lates biomolecular interactions and post-translational modifications according to its subcellular localization, was found to be down-regulated [61].
Alterations of numerous proteins have been reported in tissue aging [60, 62]. Certain proteins that are very sensitive to ROS are more prominently related to BGA (table 1).
Partial inhibition of the proteasome, a highly sophisti-cated protease complex, in young human fibroblasts accelerates cell senescence via the p53/Rb-dependent pathway, suggesting that the age-related decline in proteasome activity participates in human fibroblast senescence and, possibly, skin aging [68]. The age-related loss of proteasome activity in dermal fibroblasts is linked to the induction of matrix metalloprotease 1 (MMP-1 or collagenase-1) and decreased cell respiration [58, 69, 70]
Discussion
This literature review provides arguments to support the loss of proteostasis and proteome damage as central actors in skin aging, affecting and interacting with all other BGA hallmarks (figure 2) [71].
Aging is mainly driven by deficiencies in important cellular processes, such as DNA repair and stability, mitochondrial function, cell cycle control and apoptosis, cellular metabolism, changes in hormonal levels, and the communication between all the counterparts [72]. All these mechanisms are coordinated by the proteome. Therefore, in the skin, as in any other tissue, a damaged proteome and the resulting loss of proteostasis may be considered the first step, the root cause, of cellular dysfunction, leading to disease and aging.
In the skin, sustaining proteostasis is challenging in the context of external and internal stressors. During aging, these stressors trigger a progressive decline in the integ-rity of the proteostasis network, leading to visible signs of skin aging, such as dry and thinned skin, wrinkles and
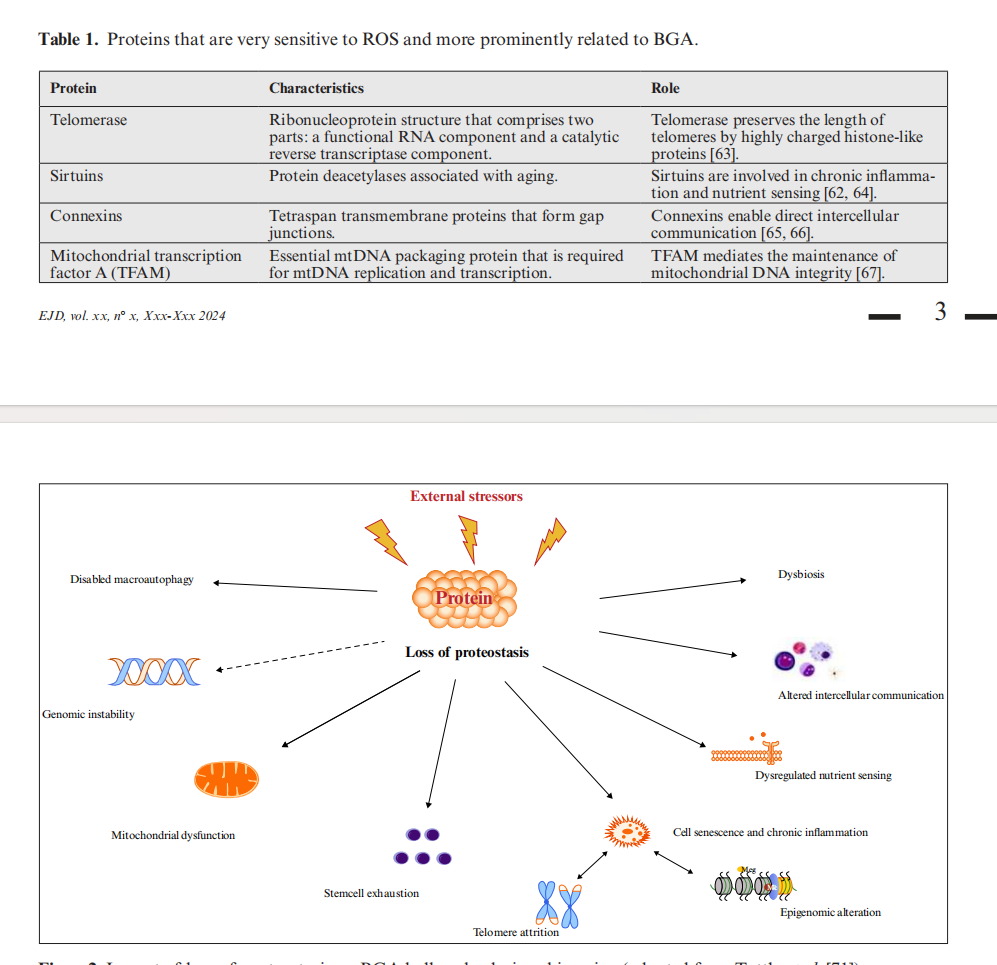
pigmentation. Therefore, the balance between protein synthesis and degradation is essential in maintaining the youthful appearance of the skin, reflecting the healthy biochemical state of cells.
Until recently, much of the research has focused on the protection and repair of DNA and genes that control and prevent cell and tissue aging. However, since proteins repair DNA and provide antioxidant protec-tion, it is no surprise that, for example, extreme resist-ance to radiation is achieved by the antioxidant protection of the proteome, rather than by effects of the genome [14]. Hence, protecting the proteome against oxidative, or other damage may be an appropriate strategy to prevent and slow down skin aging.
Interestingly, it has recently been shown that very resistant bacteria, named “polyextremophilic bacteria”, are able to survive following exposure to intense ionizing and UV irradiation, as well as hydrogen peroxide, acid, extreme temperatures and desiccation. Such robustness has been identified in relation to their capacity to safe-guard their proteome by strong antioxidant small chap-erone-like endogenous molecules, protecting proteins from carbonylation [73, 74]. Even if their DNA is exten- sively damaged, their physiology remains unaffected, which is due to highly efficient DNA repair by a well-pro-tected intact proteome, providing extreme radiation resistance. The physiology of this bacteria offers new perspectives in skin aging, opening the door to in vivo protection of the proteome against carbonylation, aggre-gation and degradation induced by external factors. Acting at the root of the cause of aging and its hallmarks should be the most effective way to mitigate the biolog-ical consequences of aging. Several therapeutic approaches have been proposed to prevent or delay skin aging. However, they usually address only one specific aging hallmark, and thus induce only a mild effect on aging. Most of them target glycation, and some carbonylation, in the skin [75-77]. Among such treatments are: plant extracts, zinc, resver-atrol derivatives and antioxidant vitamins; the aryl hydrocarbon receptor (AHR), a nuclear receptor that modulates the response to environmental stimuli and may activate innate immunity; aquaporin 3, which has been shown to reduce long-term UVA-induced senes-cence in skin fibroblasts by promoting autophagy natural antioxidants, which exhibit redox-balancing and/or iron-chelating properties; and peptides with notable effects on chronologically aged and/or photo-damaged skin [76, 78-87].
This review of the literature provides strong arguments in favour of proteome damage being the root cause of aging, indicating the importance to develop strategies to protect proteins before they are degraded. Furthermore, protecting the skin proteome from oxidative damage by small chemical antioxidant chaperones promises to miti-gate the majority of biological consequences of skin aging. Such proteome-protecting molecules exist in bacteria, accounting for their extreme radiation resist- ance. The use of such molecules in the form of a topical application to the skin represents a challenge in the management of aging, but if successful, could provide a new approach for both prevention and slowing down of skin ageing in the near future.
Conflicts of interest: Brigitte Dréno and Miroslav Radman receive funds from NAOS Les Laboratoires, France. Isabelle Benoit and Eric Perrier are employees of Naos-ILS.
Funding: this work was funded by NAOS les Laboratoires,
Data availability statement: the data that support the findings of this work are available from the corresponding author, Brigitte Dréno, upon reasonable request.
Author contribution statement: all authors contributed equally to this work.
Ethical Considerations: not applicable
Acknowledgments: the authors acknowledge the writing support of Karl Patrick Göritz, SMWS, France.



This article is excerpted from the 《Eur J Dermatol 2024》 by Wound World.

